g H 2O, must be used as a conversion factor to calculate the maximum amount of solute, ammonium iodide, NH 4I, that can dissolve in the given amount of solvent, 75.0 grams of water, H 2O. In everyday life, it is often assumed to approximate the water density to 1000 k g. 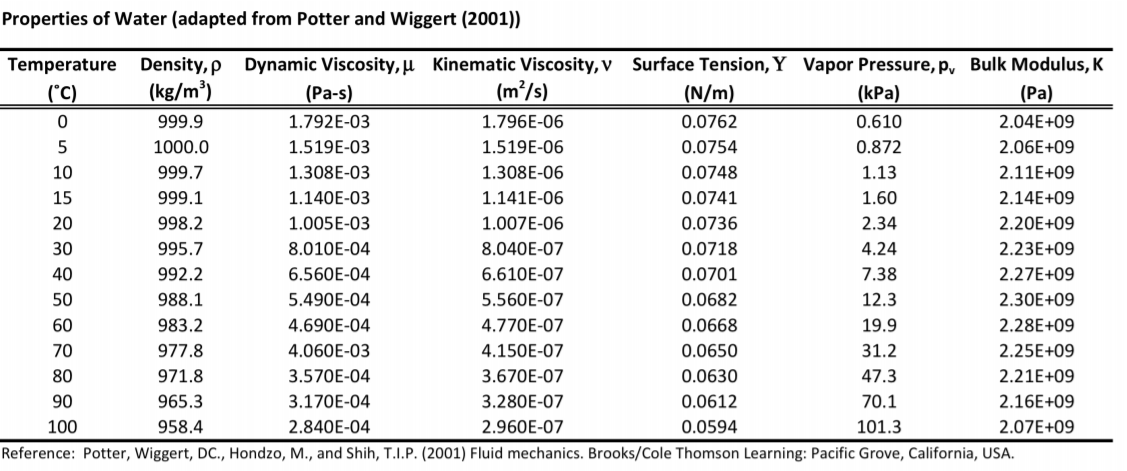
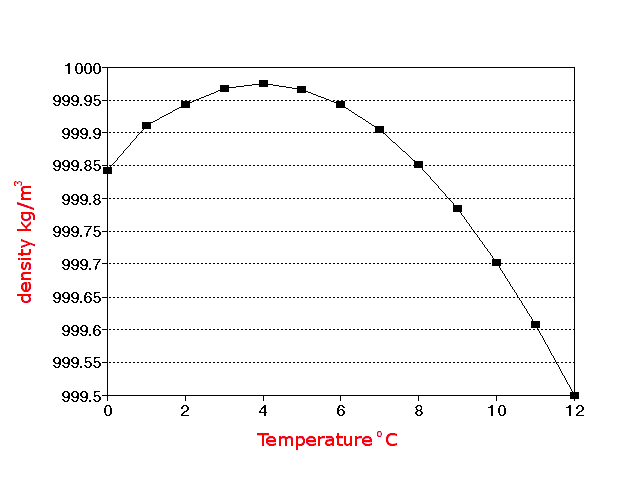
in the so-called normal conditions), the water density is equal to 1000 k g / m 3 kg/m3 k g / m 3. The density of water is approximately 1 gram/ cubic centimetre (1 g/cm3). It also changes with variations in atmospheric pressure, temperature and humidity.At 101.325 kPa (abs) and 20 ☌ (68 ☏), air has a density of approximately 1.204 kg/m 3 (0. Both below and above this temperature, the density of water is lower. The density of air or atmospheric density, denoted, is the mass per unit volume of Earths atmosphere.Air density, like air pressure, decreases with increasing altitude. So to convert celsius to kelvin, we just add 273.15 to celsius. That would be true if there were no phase change. Celsius is usually used in metric systems. In order to determine whether this solution is saturated or unsaturated, the solubility of ammonium iodide, NH 4I, which has a reported value of 172 g/100. The density of water reaches its maximum around 4☌. Water at -20 degrees Celsius heat will expand matter, so at +40 degrees Celsius, water would have less density. Most animals and plants contain more than 60 water by volume. base temperature as 60F.) Water is essential for life. 
At 4☌ pure water has a specific gravity of 1. Source: CRC Handbook of Chemistry and Physics. At 4☌ pure water has a density (weight or mass) of about 1 g/cu.cm, 1 g/ml, 1 kg/litre, 1000 kg/cu.m, 1 tonne/cu.m or 62.4 lb/cu.ft. Since the chemical formula for water, H 2O, is associated with the 100.-gram quantities in the denominators of the solubilities in Table 7.9.1, water, H 2O, is the solvent in this solution, and the remaining substance, ammonium iodide, NH 4I, is the solute, "by default." Density Of Liquid Water From 0☌ to 100☌ External pressure: 1 atm 101 325 Pa Temperature ☌ 3Density kg/m Temperature ☌ Density kg/m3 Temperature ☌ Density kg/m3 0 (ice) 917.00 33 994.76 67 979.34 0 999.82 34 994.43 68 978.78 1 999.89 35 994.08 69 978.21 2 999.94 36 993.73 70 977.63 3 999.98 37 993.37 71 977. Pure water has a density of 0.99823 grams/cubic centimeter at 1 atm pressure and a temperature of 20 degrees Celsius (68 degrees Fahrenheit). Use the solubility information that is presented in Table 7.9.1 to determine whether the resultant solution is saturated or unsaturated, and calculate the amount of excess solute that remains undissolved in this solution.īefore a solubility limit can be applied as a conversion factor, each substance that is referenced in the given problem must first be classified as a solute or a solvent. Once the system passed its tipping point, he said, Western Europe would start to cool down by as much as 3 degrees Celsius (5.4 degrees Fahrenheit) every decade. \)Ī solution is prepared by mixing 129 grams of ammonium iodide and 75.0 grams of water at 20 degrees Celsius. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
